Dissolved Oxygen
|
In water there is always a certain amount of dissolved oxygen. This is oxygen which exists as O2, not bonded to H2 to form H20. In general, the deeper the water is, the less dissolved oxygen. Dissolved oxygen is measured in milligrams per litre (mg/l), parts per million (ppm) or micromoles (umol). Fish living in saltwater require less oxygen than freshwater fish because saltwater cannot sustain as much dissolved oxygen.
Less oxygen is able to dissolve in warmer water because the higher the temperature, the more kinetic energy molecules have. So more of the intermolecular bonds break, allowing molecules to escape. This means that there are fewer cavities in the solution for O2 molecules to occupy. Increased salinity means decreased levels of dissolved oxygen in water. This is because H2O and NaCl molecules both contain polar bonds, but O2 contains non-polar bonds. Polar bonds have a greater affinity to other polar bonds than non-polar bonds. Because the H2O is bonded to the NaCl, it can’t bond so easily to the O2. |
Our experiments...
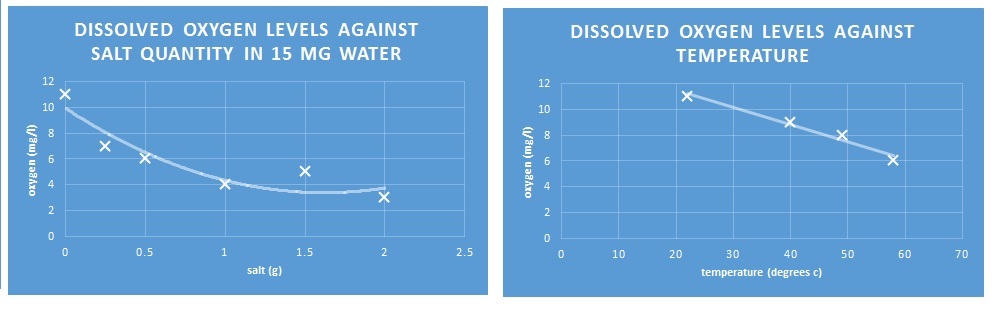
In the first experiment we measured the volume of oxygen in water of varying salinity. Each time we used 15ml of distilled water. First we added no salt, the 0.25g, then 0.5g, then 1g, then 1.5g, and then 2g. From prior research we expected water with higher levels of salt to contain less oxygen, and the general trend supported this hypothesis. However, there were some anomalies. The water with 1.5g of salt contained 5mg/l of oxygen, 1 more than that with 1g. This might be due to inaccurate measurement, but more likely due to the nature of the test we used. In order to determine the quantities of oxygen we looked at the solution next to a colour chart of varying shades of pink. Therefore, it's likely that we misinterpreted the correct shade of pink. Also, if we had added differing amounts of the colouring solution, some solutions may have had a more intense colour than others. Nonetheless, the experiment definitely did suggest that increased salinity reduces the quantity of dissolved oxygen.

In the second experiment we measured the volume of oxygen in water of varying temperatures. The process was the same as the first experiment, except that we did not add salt, but rather heat the water in water baths to different temperatures. The first was at room temperature, 22 degrees, then 40, 49 and 58 degrees. Much like with salinity, we expected higher temperatures to contain less oxygen, and our experiment confirmed this hypothesis. The volume of oxygen reduced as temperature increased.
From our experiments we can conclude that a fish would be likely to be happiest in cold freshwater since it contains the most oxygen.
For more information see: http://www.fondriest.com/environmental-measurements/parameters/water-quality/dissolved-oxygen/
https://www.boundless.com/chemistry/textbooks/boundless-chemistry-textbook/solutions-12/factors-affecting-solubility-94/gas-solubility-and-temperature-404-7627/
http://www.freedrinkingwater.com/water_quality/water-science/j-9-08-does-salt-concentration-salinity-water-affect-solubility-oxygen.htm
https://www.boundless.com/chemistry/textbooks/boundless-chemistry-textbook/solutions-12/factors-affecting-solubility-94/gas-solubility-and-temperature-404-7627/
http://www.freedrinkingwater.com/water_quality/water-science/j-9-08-does-salt-concentration-salinity-water-affect-solubility-oxygen.htm